Mesoblast Limited (ASX MSB; Nasdaq MESO) is a Melbourne-based leading global stem cell company focusing on R&D in regenerative cellular medicines. The Mesoblast share price has been falling since 2011 but has since stabilised in the $1.20 to $2.00 range in the past two years.
Despite the company burning piles of cash each year, investors are optimistic about the prospects of MSB and have allowed the company to access fresh financing in support of its R&D activities. As a result, revenue increased to US$14.9 million from US$0.6 million in the previous corresponding period, thanks to earnings from two licenses marketing Mesoblast products in Japan and Europe.
However, greater regulation among different countries, ethical issues, the potential for more capital rising due to multiple ongoing costly clinical trials and uncertainty in commercialising their products posts risks to the company.
About Mesoblast Limited (ASX MSB; Nasdaq MESO)
Mesoblast (ASX MSB), as a cellular medicine company using stem cells to treat a myriad of diseases and ailments including inflammatory ailments, cardiovascular disease and back pain is being touted as the next CSL (ASX CSL). Mesoblast is listed on the ASX and Nasdaq with a market cap of AU$1.62bn.
MSB’s market-leading position combined with high capital requirements form a natural barrier that prevents competitions from new entrants in Australia. However, there are two emerging companies in the Australia market, Cynata Therapeutics (ASX: CYP) and Regeneus (ASX: RGS) which will compete in this space.
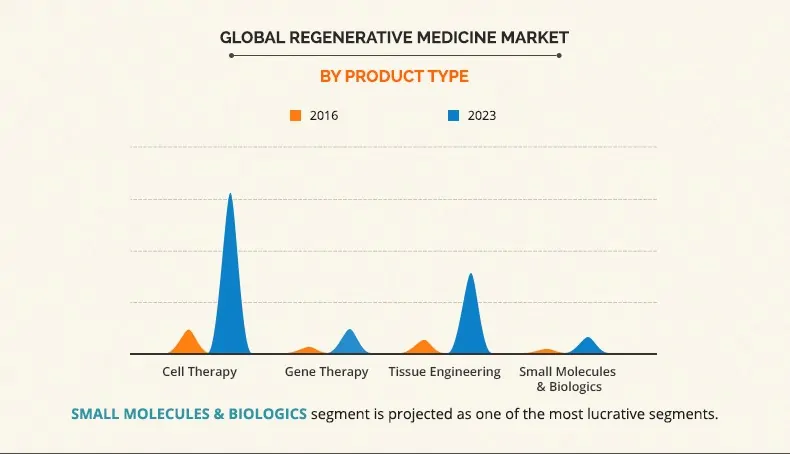
Emerging Global Regenerative Medicine Market Worth $32 Billion in 2023
According to Global Regenerative Medicines Market – Analysis and Forecast report, the regenerative medicine market was valued at $5.44 billion in 2016 and is expected to reach $32.325 billion by 2023, growing at a rate of 32.2% from 2017 to 2023.
Based on product type, the market is segmented into cell therapy, gene therapy, tissue engineering, and small molecules & biologics. Moreover, among all the regenerative medicine products, the cell therapy segment has the most promising future due to its efficiency to restore lost function of tissues and organs. This offers phenomenal opportunities for the growth of Mesoblast.
Burning Cash But Close To Commercialisation
The biggest problem with Mesoblast is their huge cash outflows and historic lack of revenue. At end of H1 2018, MSB posted US$35.2 million operating expense that is mostly attributed to employee salary and multiple clinical trial expenses.
Based on the Q3 FY2018 quarterly report, the approximate cash burn is US$6.52m per quarter. Considering the US$50M Financing with NovaQuest Capital in July 2018, the total cash on hand is approximately US$109.54m. Therefore, MSB has the ample funding to support its operational activities for at least the next 17 months.
However, Mesoblast is now moving towards a stage in its development where it has started commercialising some of its product candidates.
MSB was given an award by US-based consultancy Frost & Sullivan on December 2017, as an international industry leader due to its cutting-edge mesenchymal lineage cell technology platform. It now has four tier one cell therapy product candidates for the treatment of acute Graft versus Host Disease (aGVHD), chronic heart failure, chronic lower back pain and chronic diabetic nephropathy.
Mesoblast also has three tier two product candidates including MPC-75-IA (Knee Osteoarthritis), MPC-25-IC (Acute Cardiac Ischemia) and MPC-25-Osteo (Spinal Fusion).
Moreover, MSC-100-IV, as one of tier one product candidates was licensed to Japanese JCR Pharmaceuticals Co. Ltd for the treatment of aGVHD in 2015. It has already acquired approval from the Japanese government and is now sold in the Japanese market. In HY18, around $1.6 million in revenue from this has been recognized. Just recently, this stem cell medicine has also passed Phase 3 trials. If this product can secure approval from the U.S. healthcare regulator, the Food and Drug Administration, aGVHD will be sold in the lucrative US market commercially.
In addition, MSB granted TiGenix NV (most advanced cell therapy platform in Europe) with an exclusive right to sell mesenchymal stem cell product named Cx601 for the treatment of fistulae, including Crohn’s disease. As consideration, MSB received up to US$24 million in payments and single-digit royalties on net sales of Cx601.
Alliance with China’s top Pharmaceutical Group Tasly
On 17 July 2018, MSB entered into a strategic alliance with one of the largest pharmaceutical groups in China, Tasly, for the MPC-150-IM (for prevention of chronic heart failure) and MPC-25-IC (for prevention of acute myocardial infarction) product candidates. MSB will receive US$40 million on closing, comprised of US$20 million upfront technology access fee and US$20 million in an equity purchase in MSB at $1.86 per share. MSB will also receive US$25 million on product regulatory approvals in China and double-digit escalating royalties on net product sales. MSB is eligible to receive six escalating milestone payments upon the product candidates reaching certain sales thresholds in China. This transaction is subject to governmental approvals from China.
The considerable size and volume of the cardiovascular market in China represent a significant opportunity for MSB and Tasly. For instance, 17% of the 1.3 billion people there have cardiovascular disease. 500,000 individuals per year experience cardiac arrest, presumably due to various factors, including substandard air quality, smoking, excessive salt intake and inactive lifestyles. It is estimated that the cardiovascular disease drug market in China will rise to $45 billion in 2018.
Regulatory and Market Approval An Obstacle for the Commercialisation
The first step to bringing the product candidates to market is the acquisition of the license or collaboration agreement with existing product candidates. If not, MSB must delay or discontinue the development of the product candidates, which in turn decreases the MSB ability to generate profit and support the operation.
There are several reasons that could cause the failure of the obtaining the regulatory approval. Firstly, the regulatory approval process is expensive, time-consuming and unpredictable. Obtaining market approval can take several years, depending on the country, the quality of the clinic data necessary to gain approval, and procedures of the reviewing regulatory authority. Even then, there is no guarantee that the regulatory approval will be granted due to numerous factors, including the discretion of the regulatory authorities.
Secondly, the government regulators may disagree with the product candidate’s usage. The cellular therapy product candidates, which are used to treat human illness, are subject to extensive legislation. Different government authorities around the world have various attitudes towards the products, which will be reflected in the administration and enforcement of the related laws and regulations. Take the approval process in the United States, for example, the company has made an effort to apply for breakthrough therapy designation for the product candidates, which is supposed to accelerate the approval process under conventional FDA (the United State Food and Drug Administration) procedures. Even if the company believed the products candidates meet the criteria as the therapy designation, FDA may disagree. Furthermore, even if FDA agrees on the designation as a breakthrough therapy, it does not ultimately guarantee market approval.
Finally, ethical and social concerns related to the use of the embryonic stem cells (“ESCs”) could negatively affect the public’s perception of the company’s product candidates, which could harm the MSB business. MSB claims its cells are not ESCs and its product candidates, such as MLCs, are frequently misunderstood by the public. The lack of the understating of the difference between ESCs and non-ESCs could, in general, results in the negative impact on the company.
Revenues Showing Growth
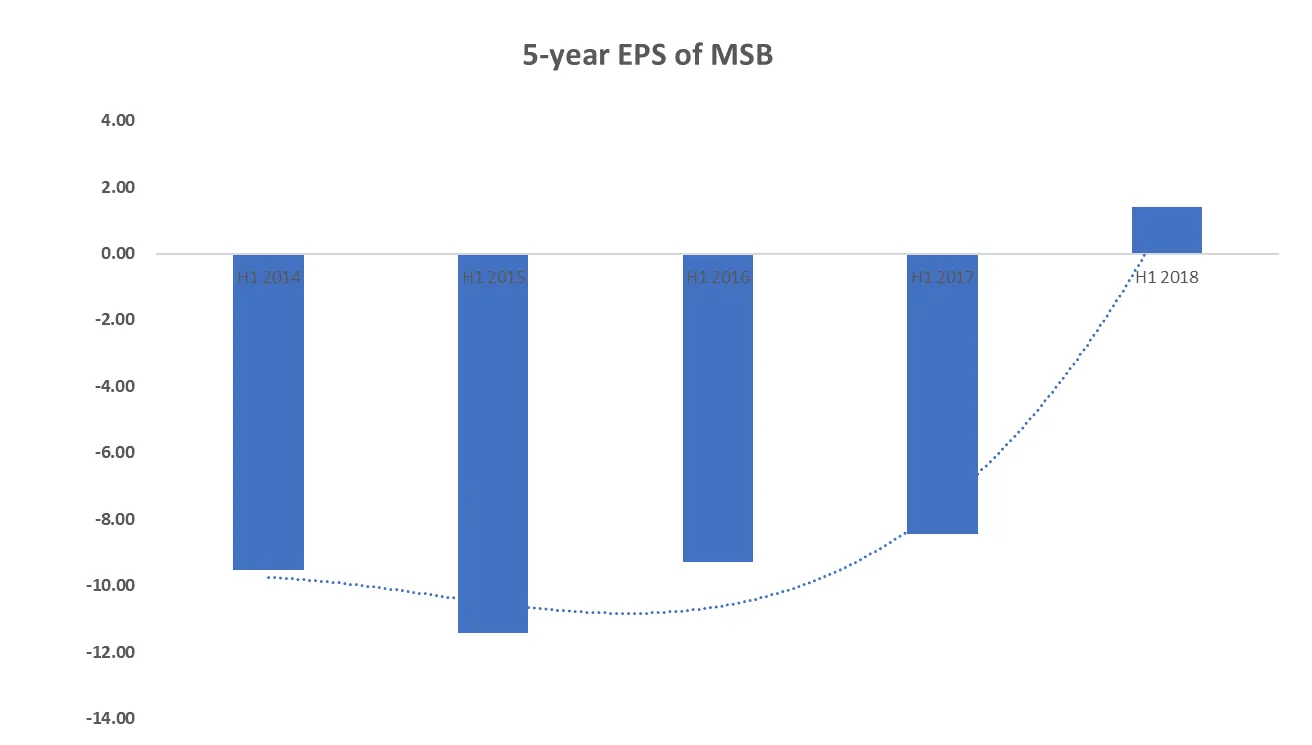
Over the last five years, Mesoblast shares revenue growth has been stagnant but grew strongly in H1 2018. H1 2018 revenue was US$14.6 million, up US$13.7 million compared to the corresponding period in 2017. The two main contributors to the growth are the sales of the TEMCELL® Hs. in Japan and the execution of the patent licence agreement with TiGenix.
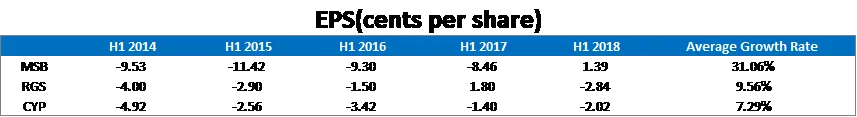
In H1 2018, MSB is the only company with positive earnings out of its peers, Cynata Therapeutics (ASX: CYP) and Regeneus (ASX: RGS), at US 13.7 million, which is in line with its strong growth in revenue. On a per share basis, Mesoblast shares EPS has turned positive in H1 2018.
Mesoblast Shares Is A High-Risk Speculative Play
Mesoblast shares is a high-risk play, given the number of capital raisings it has conducted historically, lack of revenue and its high cash burn due to R&D activities. The biotech space is still a high-risk part of the market and is further challenged by stronger regulations and ethical-related problems.
Despite a lot of downside risk, Tasly clearly sees a lot of promise in the candidates and must be reasonably confident that they will end up coming to market. A strategic alliance with Tasly Pharmaceutical Group helps investor confidence with Mesoblast’s ability to commercialise these products.
The promising aspects of the regenerative medicine market could be the next big thing and if Mesoblast can successfully bring to market several of its products, Mesoblast shares can have very high upside potential.